METHODS
How the analysis was done
Sample preparation
UC tissue was harvested from 11 single-source, healthy equine donors. Cryopreserved at −80 °C and processed into microparticulate suspension. A total of 13 samples were analyzed.
Proteomic analysis
High-resolution tandem mass spectrometry (Orbitrap Fusion Tribrid) was used. Protein identification and relative quantification were based on total spectral counts (TSC). Proteins were annotated using the Equus caballus proteome and UniProt databases.
Cytokine quantification
ELISA was performed on the UC suspension (non-particulate fraction). Analytes included IFNg, IL-1a, IL-1ra, IL-2, IL-4, IL-8, IL-10, IL-15, MCP-1, VEGF, and hyaluronic acid (HA).
RESULTS
Proteomic findings
+2,600 proteins were identified and annotated. 80 proteins had high abundance (TSC ≥ 100) and were consistently present across all donors. Proteins with TSC ≥ 40 were considered functionally relevant and included structural proteins (e.g., fibrillar collagens, glycoproteins), regulatory proteins (e.g., clusterin, cadherin), and anti-inflammatory and anabolic mediators (e.g., IGF-2, DDAH2). Triplicate samples from a single donor showed a mean coefficient of variation (CV) of 4.7% for high abundance proteins, indicating high batch consistency.
Cytokine profile
Inflammatory cytokines (IL-1a, IL-2, IL-4, IL-8, IL-15, IFNg) were low or near the detection limit. Anti-inflammatory cytokines (IL-1ra, IL-10) and growth factors (VEGF, HA) were consistently detected. The IL-1ra/IL-1a ratio averaged 0.3, suggesting a favorable anti-inflammatory baseline. CVs for cytokine measurements were low (e.g., IL-1a 1.1%, MCP-1 3.8%).
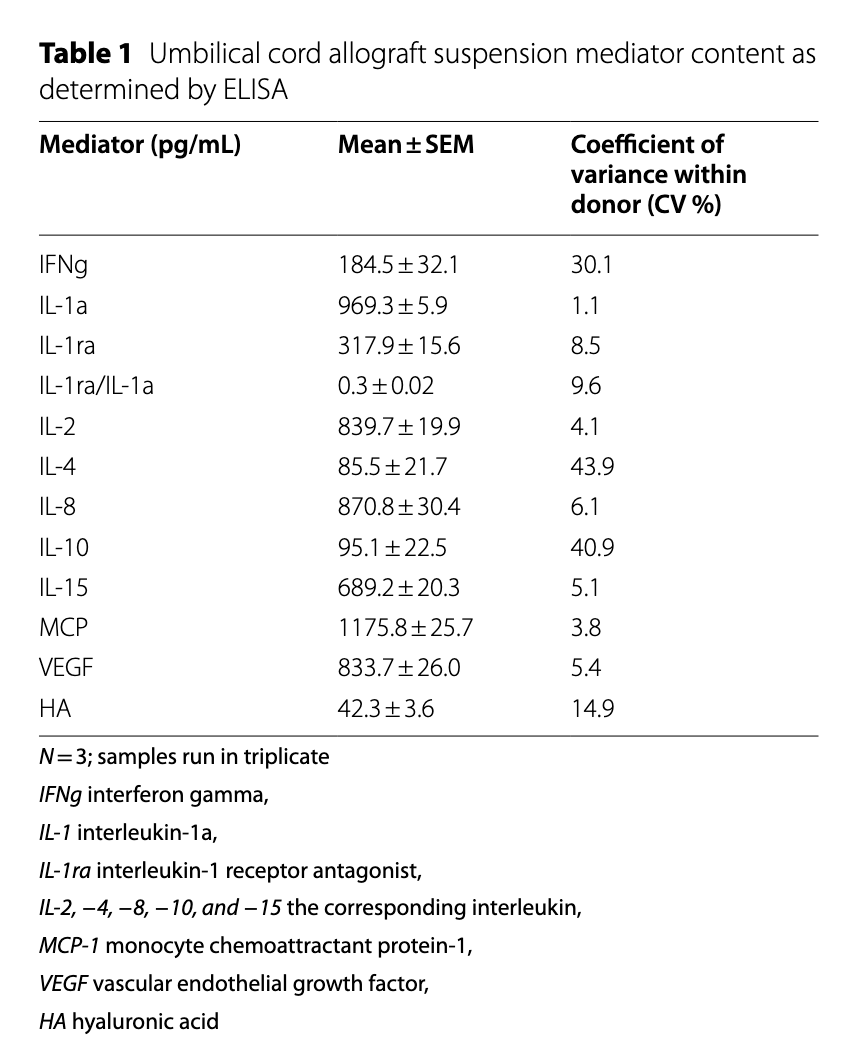
Table 1 — Umbilical cord allograft suspension mediator content as determined by ELISA. N=3; samples run in triplicate.
CONCLUSION
What the data tells us
The in vitro analysis demonstrated that the cryopreserved equine UC allograft is protein-rich, biologically active matrix with consistent composition across donors and batches. The low inflammatory cytokine content and presence of anti-inflammatory and regenerative mediators support its potential as a safe and effective biologic scaffold for musculoskeletal tissue repair.
In Vitro Findings Summary
| Category | Key Finding | Relevance |
|---|---|---|
| Protein Consistency | +2,600 proteins identified; 80 highly abundant proteins consistently present across all donors | Demonstrates batch-to-batch consistency and product reliability |
| Protein Functionality | Includes structural (collagens, glycoproteins) and regulatory proteins (IGF-2, DDAH2, clusterin) | Supports tissue repair, anti-inflammatory action, and cellular homeostasis |
| Low Inflammatory Cytokines | IL-1a, IL-2, IL-4, IL-8, IL-15, IFNg low or near detection limits | Indicates low immunogenicity and a safe injection profile |
| Anti-inflammatory Cytokines | IL-1ra and IL-10 consistently detected; IL-1ra/IL-1a averaged 0.3 | Highlights anti-inflammatory potential and therapeutic relevance |
| Growth Factors | VEGF and hyaluronic acid present in all samples | Supports angiogenesis and joint lubrication for tissue regeneration |
| Batch Variability | Triplicate samples from one donor showed mean CV of 4.7% | Ensures manufacturing precision and reproducibility |
| Cytokine Stability | Low coefficient of variation for cytokines (e.g., IL-1a 1.1%, MCP-1 3.8%) | Reinforces product stability and quality control |
Ready to explore equicenta® CTM for your practice?
The peer-reviewed data speaks for itself: consistent protein profiles, proven biocompatibility, and measurable clinical outcomes in musculoskeletal conditions.
Get More Info
