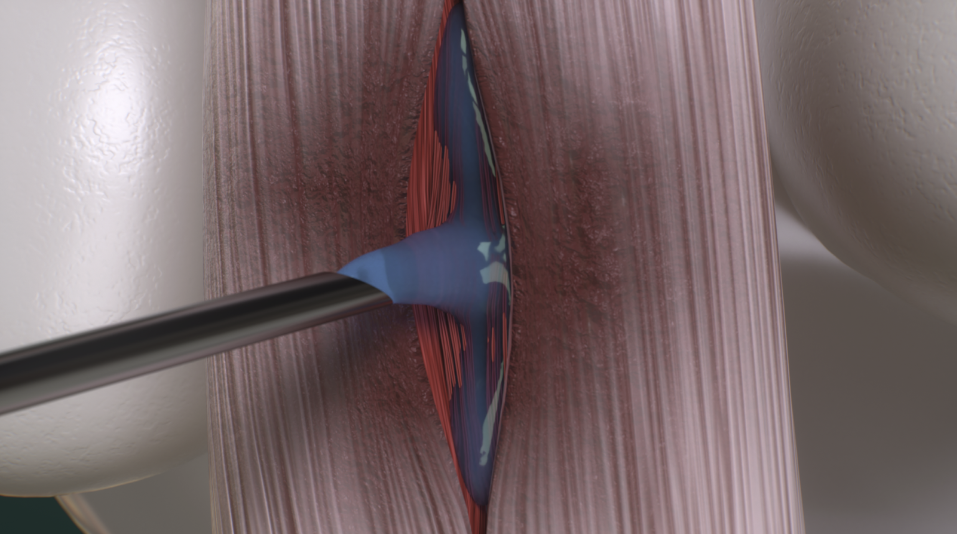
Joint Treatment
Support joint health with a connective tissue matrix designed to complement your veterinarian’s treatment plan. equicenta® CTM provides a scaffold and bioactive signals within the joint environment, helping support comfort, function, and long-term soundness—without relying on steroids or repeated procedures.
See How It Works