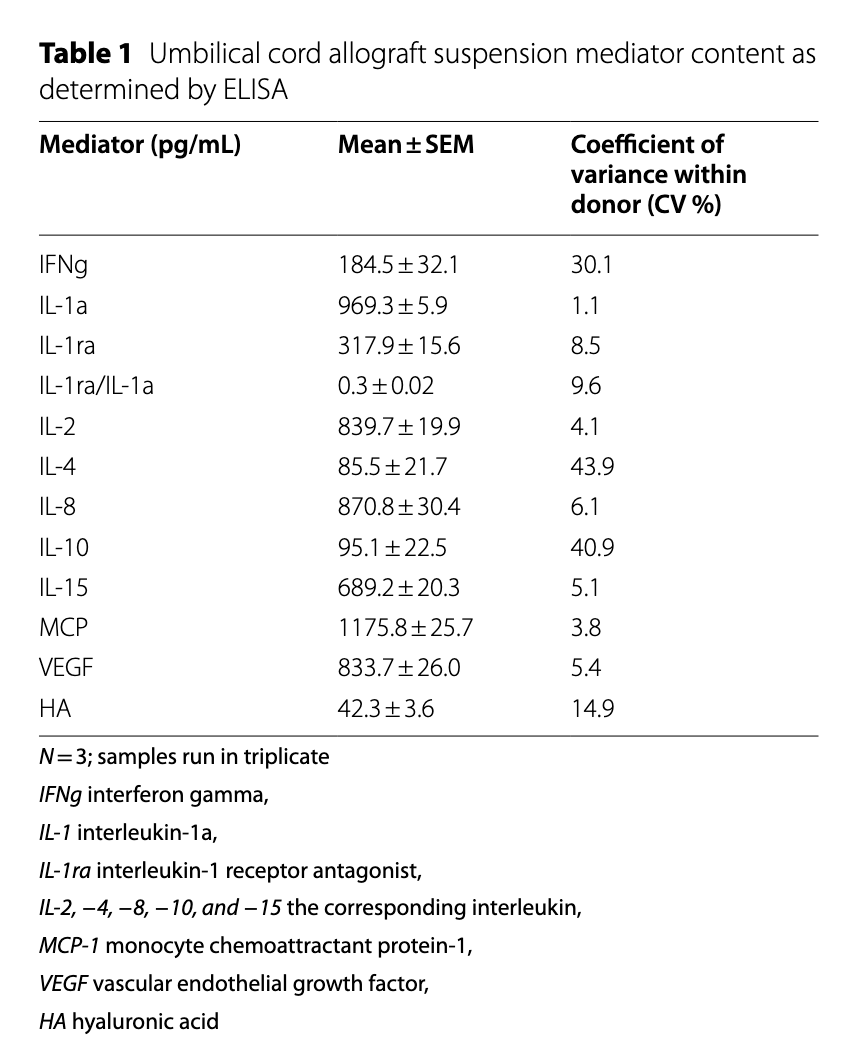
Table 1 — Umbilical cord allograft suspension mediator content as determined by ELISA. N=3; samples run in triplicate.
STUDY OVERVIEW
Objective & methods
Objective
To evaluate the consistency of protein content and cytokine profile in cryopreserved UC allograft.
Methods
Mass spectrometry (MS) and ELISA were used to assess protein abundance and cytokine levels in samples from 11 donors.
RESULTS
Proteomic consistency across donors and batches
- A total of +2,600 proteins were identified. Of these, 80 proteins exhibited high abundance (TSC ≥ 100), and 224 proteins showed moderate abundance (TSC ≥ 40), with a coefficient of variation (CV) of 16.1%.
- Proteins with TSC ≥ 40 were consistently present and functionally relevant (e.g., collagens, glycoproteins).
- Triplicate samples from a single donor showed <5% coefficient of variation in abundance proteins, indicating high batch consistency.
- Functions included structural support, antioxidant protection, and regulation of inflammation.
ELISA cytokine consistency
- Cytokine levels (e.g., IL-1ra, IL-10, VEGF) were measured in UC suspension.
- Inflammatory cytokines (e.g., IL-1, IL-2, IL-4) were low or near detection limits.
- Anti-inflammatory markers (IL-1ra, IL-10) and growth factors (VEGF, HA) were consistently present.
- Low variability across triplicates confirmed reproducibility.
CONCLUSION
What the consistency data tells us
The cryopreserved UC allograft demonstrated a highly consistent protein and cytokine profile across donors and batches. These findings support its reliability and reproducibility as a biologic scaffold for musculoskeletal applications.
Reference
1 Bertone, AL et al. “Cryopreserved equine umbilical cord tissue allograft characterization and biocompatibility in vivo in musculoskeletal tissues: a controlled study.” BMC Medicine, 2025.
Ready to explore equicenta® CTM for your practice?
The peer-reviewed data speaks for itself: consistent protein profiles, proven biocompatibility, and measurable clinical outcomes in musculoskeletal conditions.
Get More Info
