Our Team
While others chased speed, we chose evidence. Equine Performance Labs has spent its years building the science before making the claim — controlled studies, peer-reviewed publication, and a standard of rigor borrowed from human medicine. One principle has held throughout: Horse First. Sport Second.
Read Our StoryAll About Our Story
Heather Rogers built Equine Performance Labs as a horse girl with a foundational knowledge of human biologics. She knew the equine market wasn’t offering what the science could deliver — so she founded a company that would. EPL was built on a single belief: horses deserve better. Better quality. Better science. Better solutions. What started as a conviction became a mission rooted in research, data, and a commitment to advancing equine care through science-backed innovation.
Meet Our Team


Our Story
A quiet build, by design. Here’s how the work has unfolded.
The work starts with a conviction — that horses deserved options the market wasn’t offering, and that the rigor applied in human biologics could be brought to equine care. By June, our CEO Heather Rogers begins forming the founding team, drawing together veterinary surgeons, biologics scientists, equine breeders, and operational leaders. The hypothesis takes shape, and the foundational work begins: building a product that could meet the rigor of human-quality compliance — for horses.

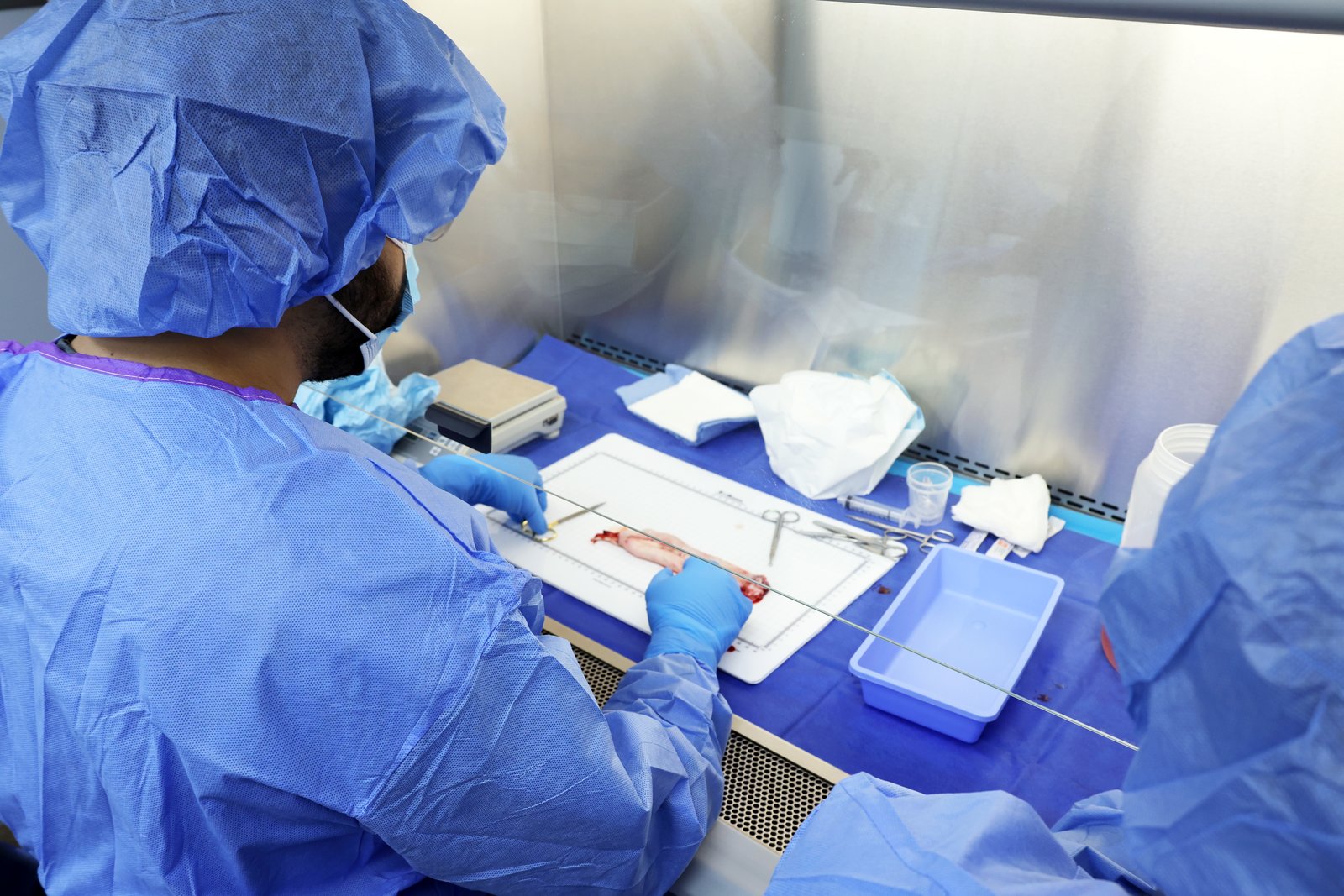
Foaling season at Coteau Grove Farms — a closed-herd thoroughbred breeding farm in Louisiana — and the partnership goes operational. The first umbilical cord is collected, and a year of theory and benchwork meets its first real test: can the team produce what they had been planning? Coteau Grove’s closed, single-source herd anchors sourcing, welfare, and traceability from this point forward.

The first finished material is produced. The work of the prior year holds — but the team knows the harder work is just beginning.
The protocol for the first safety study is approved and the study formally begins — a pivotal moment for both the company and the product.
Partway through the initial safety study, the data made something clear: the work needed more. Rather than push forward with a study we weren’t fully confident in, the team paused, returned to the bench, and rebuilt — refining the process and protocol to a higher standard than where we had started.
From that refinement, the redesigned clinical trial is initiated — the study that would later be published in BMC Medicine. The work demanded more, and so the team grew. The processing team expands, operational systems mature, and every step is rebuilt to be repeatable, traceable, and auditable.
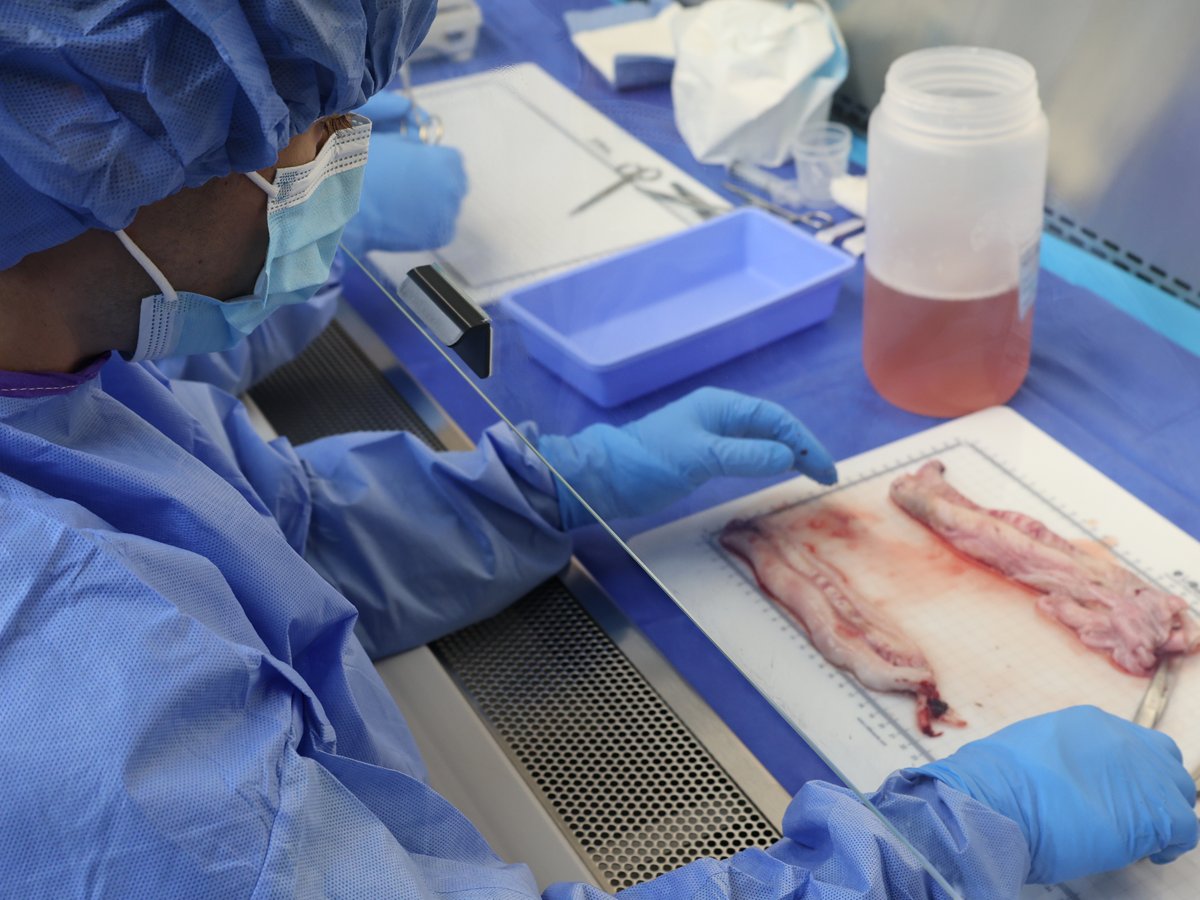
The refined safety study—built to a higher standard after the 2024 redesign—is published in BMC Medicine and indexed on PubMed. The publication reflects the level of consistency and quality the team has held to from the beginning. No adverse events were reported in the study population.
The clinical trial reaches full enrollment, marking the transition from study build-out to study execution.
Equine Performance Labs presents equicenta® CTM — the first-in-class equine umbilical cord tissue allograft — at AAEP. After three years of head-down work, the team steps into the broader veterinary community to share, educate, and listen.

The company’s quality management system is formally certified to ISO 13485:2016 — a globally recognized standard for medical device quality systems.
The final clinical trial data point has been collected. Data analysis and study reporting are now being prepared. Study to launch soon. Click here to get updates and we’ll let you know the moment it’s out.
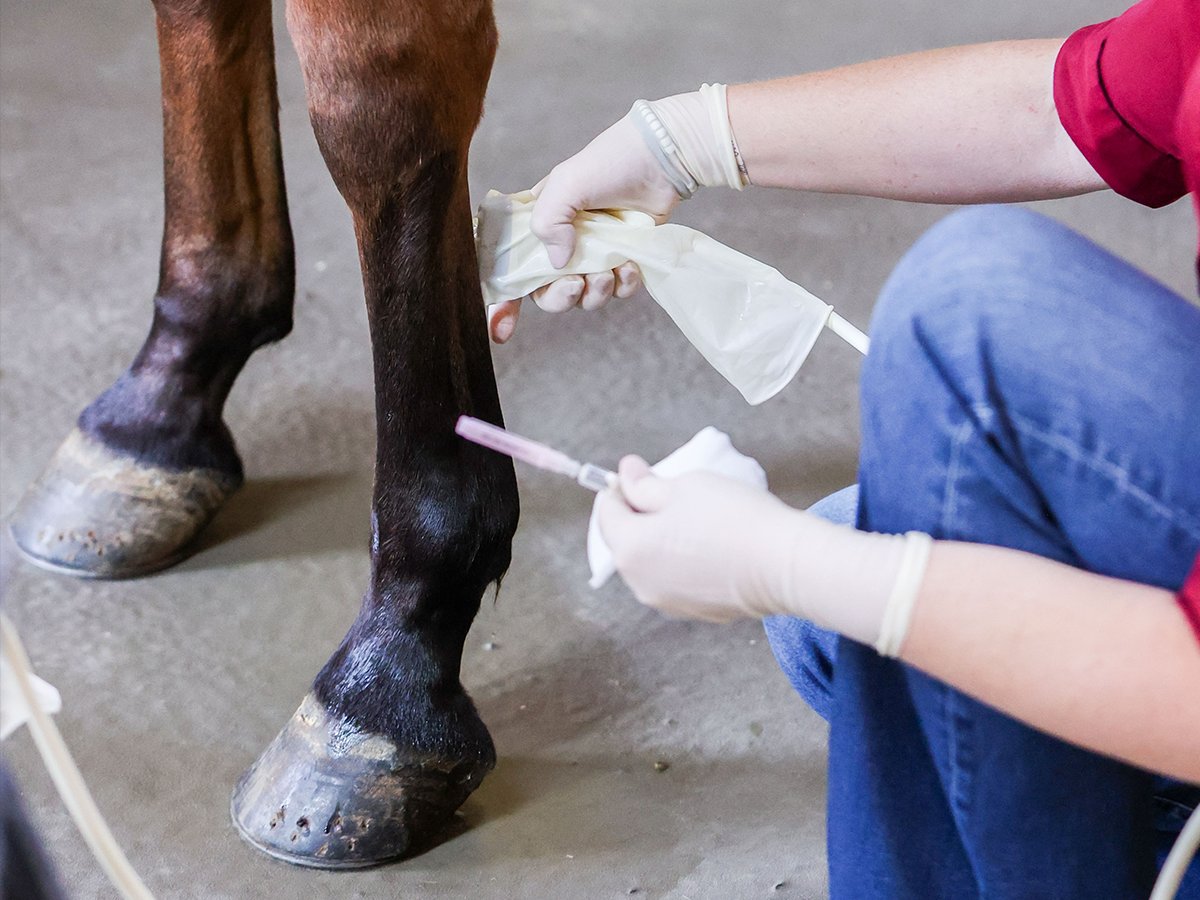
“The horse’s well-being comes before performance outcomes. Performance is a byproduct of proper care.” The Founding Principle
Meet Our Team
Veterinary surgeons, equine breeders, biologics scientists, and operational leaders. Built around one starting point.

Chief Executive Officer
Equine Performance Labs

Chief Operating Officer
Equine Performance Labs

Independent Clinical Consultant

Director of Operations
Equine Performance Labs

Business Development Director — East
Equine Performance Labs

Business Development Director — West
Equine Performance Labs

Director of Quality
Equine Performance Labs

Director of Marketing + Design
Equine Performance Labs

Partner
Equine Performance Labs / Coteau Grove Farms

Director of Operations
Coteau Grove Farms
We partner with veterinarians, clinics, and organizations who share the principle that put us here. Reach out — we’d like to hear from you.
Get in Touch